Bispecific antibody-drug conjugates (ADCs) targeting dual tumor-associated antigens (TAAs) offer advantages in fighting against cancer. These include
-
the ability to simultaneously target multiple tumor-driven proteins, potentially overcoming drug resistance.
-
recognize cells co-expressing both targets to increase tumor specificity and reducing off-target toxicity.
-
the combination of two targets increases the internalization and thus tumor killing.
-
Improving the enrichment and exposure of ADCs in tumor tissues for stronger and longer lasting killing of heterogeneous tumors
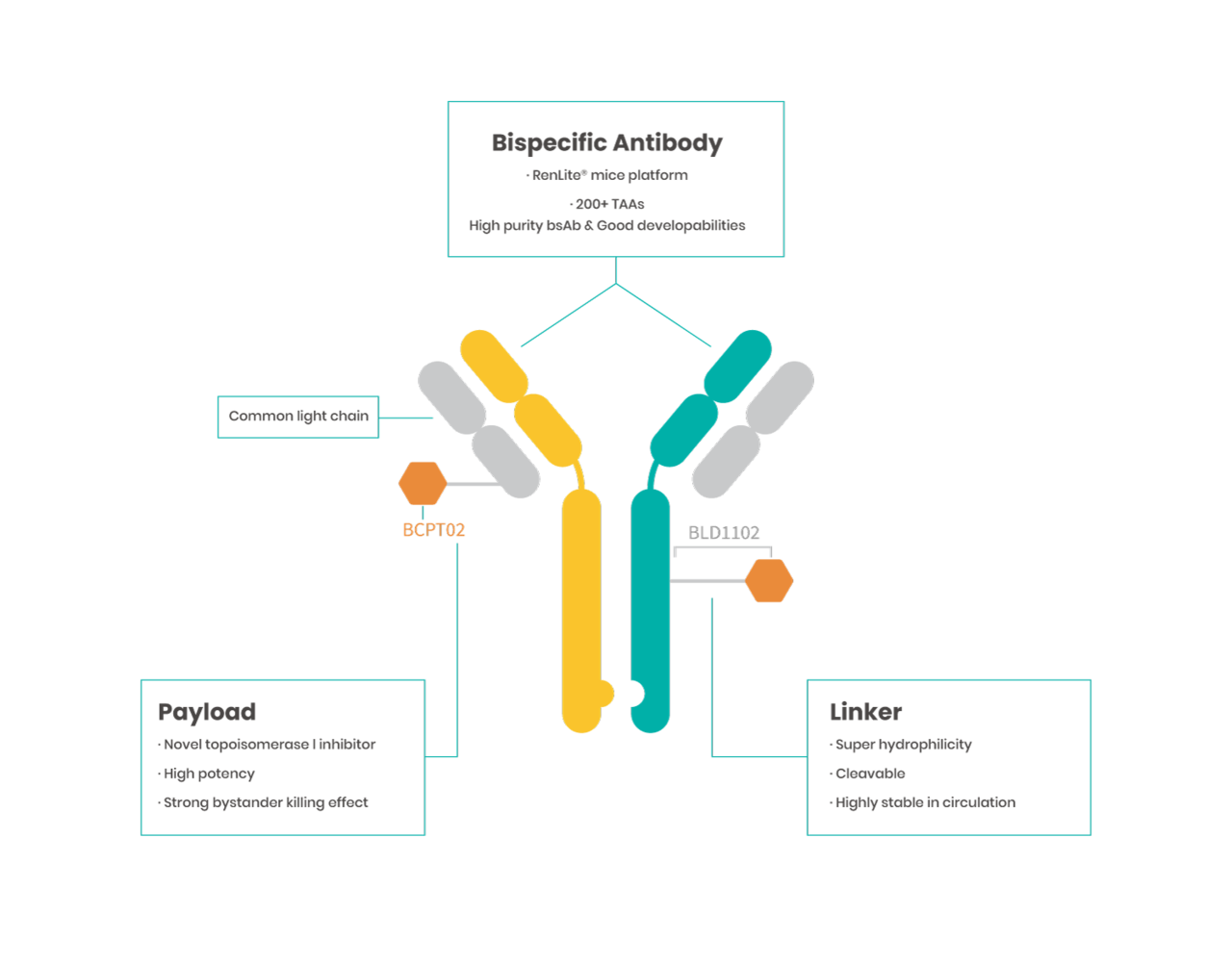
Advantages of Biocytogen’s novel bsADC platform
-
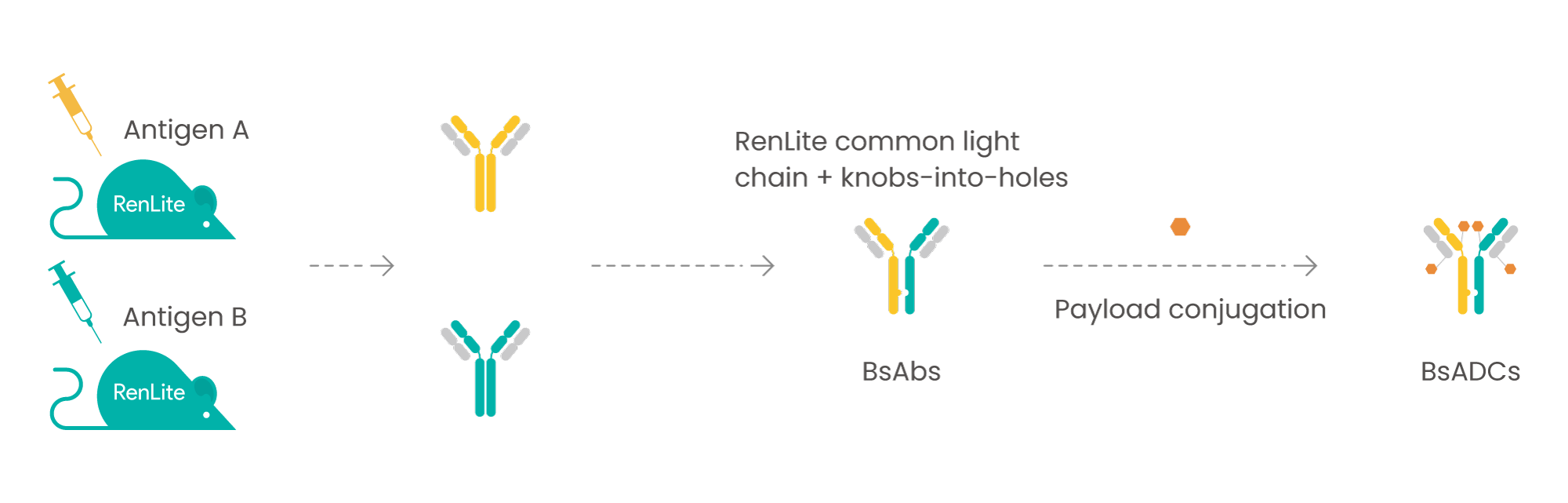
RenLite-derived common light chain antibodies are easy to assemble into BsAb with low mismatch rate and ideal physiochemical properties.
-
Combined with target gene knockout strategy, RenLite KO mice can generate antibodies with increased diversity, potentially against novel epitopes and with cross-species reactivity.
-
Proprietary linker/payload system BLD1102
-
Rapid generation of high-quality BsAbs/BsADCs for high-throughput in vitro and in vivo screening
Novel linker/payload system BLD1102 independently developed by Biocytogen
Briefly, BLD1102 is a new ADC platform developed by Biocytogen independently.
-
BCPT02 is a novel topoisomerase 1 inhibitor with broad-spectrum and potent tumor killing ability.
-
The uniquely designed cleavable linker has excellent hydrophilicity, making ADCs as hydrophilic as monoclonal antibodies, greatly improving ADC’s physiochemical properties.
-
BLD1102-ADCs exhibit good stability in plasma and more potent tumor killing compared to vcMMAE.
-
BLD1102 demonstrated good tolerabilities in preclinical non-human primates.
Biocytogen’s RenLite-based BsADC platform
-
BsADC developed from RenLite mice
Biocytogen's RenLite-based BsADC platform video
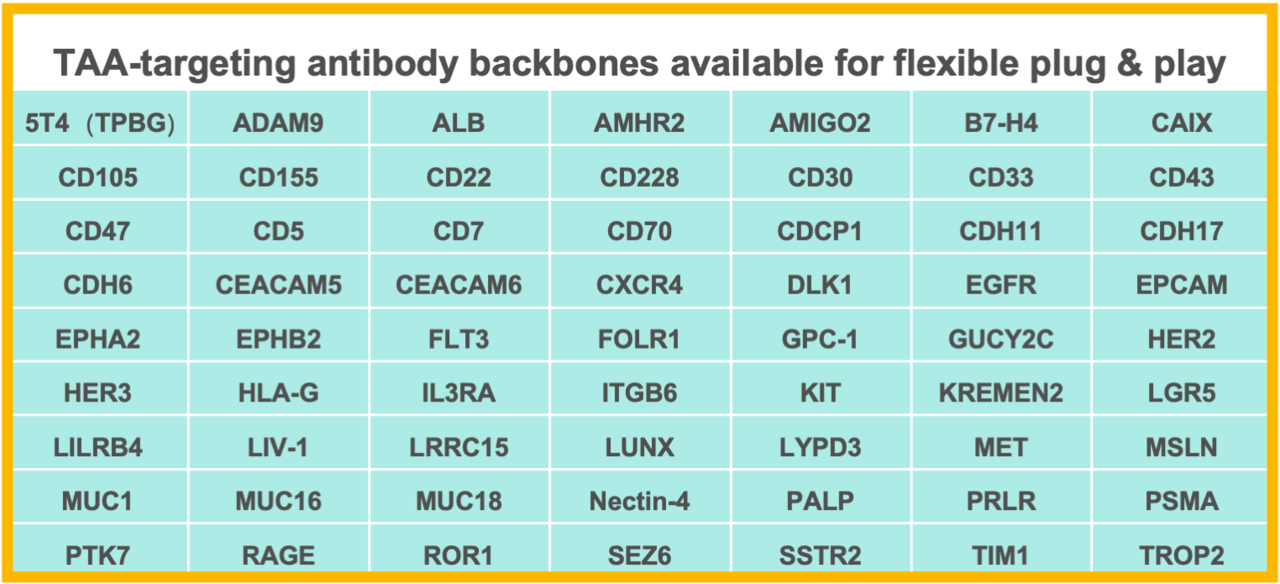
Off-the-shelf TAA-targeting antibodies available for flexible plug & play
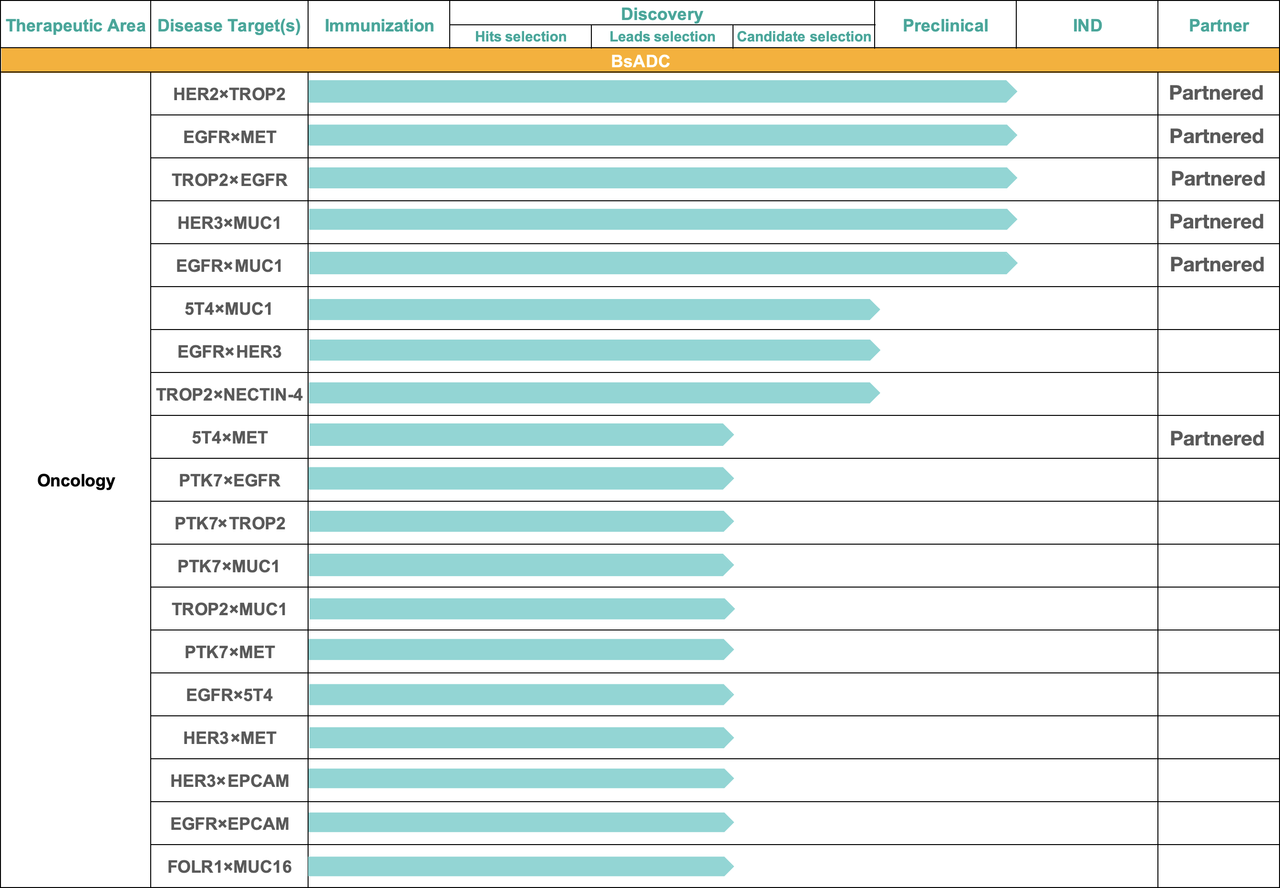
20+ BsADC programs available for partnership
Poster
SITC 2023: *DM001, a Novel TROP2xEGFR Bispecific ADC, Demonstrates Potent Tumor Growth Inhibition in Preclinical Models and Favorable Safety Profile in Cynomolgus Monkey
SITC 2023: Preclinical Evaluation of Fully Human Bispecific Antibody-drug Candidates Targeting HER3 and the Juxtamembrane Region of MUC1
SITC 2023: *DM005, an EGFR х MET Bispecific Antibody-drug Conjugate With a Novel DNA Topoisomerase I Inhibitor Payload, Showed Robust Anti-tumor Activity in Preclinical Models
SITC 2023: BSA01, a Bispecific Antibody-drug Conjugate Targeting EGFR and Membrane-bound MUC1-C, Exhibits Anti-tumor Efficacy In Vivo Efficacy Against Triple-negative Breast Cancer Xenografts
SITC 2023: BCG022: A HER3×MET Bispecific Antibody-drug Conjugate (BsADC) Targeting Key Mechanisms of Bypass Resistance in Multiple Tumor Types
SITC 2023: A Novel Bispecific Antibody-drug Conjugate Targeting PTK7 and TROP2, BCG033, Demonstrates Preclinical Efficacy Against Triple-negative Breast Cancer Xenografts
AACR 2023: A First-In-Class Anti-HER2/TROP2 Bispecific Antibody-Drug Conjugate (YH012) Exhibits Potent Anti-Tumor Efficacy
AACR 2023: YH013, a Novel Bispecific EGFR x MET Antibody-Drug Conjugate, Exhibits Potent Anti-Tumor Efficacy
AACR 2023: A Novel EGFR x MUC1 Bispecific Antibody-Drug Conjugate, BSA01, Targets MUC1 Transmembrane Cleavage Products and Improves Tumor Selectivity
AACR 2023: BCG022: A Novel Bispecific Antibody-Drug Conjugate Targeting HER3 and MET
AACR 2023: Discovery of BCG033, A Novel Anti-PTK7 x TROP2 Bispecific Antibody-Drug Conjugate with Promising Efficacy Against Triple-Negative Breast Cancer
AACR 2023: A First-In-Class Anti-TROP2/EGFR Bispecific Antibody-Drug Conjugate, DM001, Exhibits Potent Anti-Tumor Efficacy
AACR 2023: A First-In-Class Bispecific Antibody-Drug Conjugate (DM002) Targeting HER3 and the Juxtamembrane Domain of MUC1
AACR 2023: Identification of DM004, A First-In-Class Anti-5T4/MET Bispecific Antibody-Drug Conjugate
IO Summit EU 2023: YH013, a Common Light Chain Bispecific ADC Targeting EGFR and MET, Improves Preclinical Efficacy Over Its Parental Single-Targeting ADCs
AACR 2022: YH012, a Novel Bispecific Anti-HER2 and TROP2 Antibody-Drug Conjugate, Exhibits Potent Antitumor Efficacy
World ADC San Diego2022:YH013, a fully-human EGFR x MET bispecific ADC exhibits outstanding specificity and anti-tumor efficacy
PEGS-EU 2022:YH013, a fully-human EGFR x MET bispecific ADC exhibits outstanding specificity and anti-tumor efficacy