Psoriasis Model Introduction
Psoriasis is a type of skin disease characterized by hyperproliferation of epidermal keratinocytes. It is thought to be mediated by cells and molecules of both the innate and adaptive immune systems, as a result of a combination of genetic, and environmental influences. The most common model of skin injury and inflammation used in preclinical studies is an imiquimod (IMQ) -induced skin injury and inflammation model, which has phenotypes similar to human psoriasis. IMQ causes skin damage in mice, including rash and desquamation, thickening of the skin epidermis, and histopathologically visible dermis dominated by hypokeratosis and inflammatory leukocyte infiltration. IL-17 cytokine levels are significantly increased in the IMQ-induced psoriasis model, which is considered particularly important in psoriasis pathogenesis.
Biocytogen constructed a series of IMQ-induced psoriatic mouse models for preclinical studies and pharmacodynamic evaluation of psoriasis-related drugs targeting human cytokines.
Establishment of Psoriasis Mouse Model
Experimental Animals:C57BL/6 or Balb/c, 8 weeks old, female
Modeling reagent:IMQ
Modeling method: Topically applicated on dorsal for consecutive 5-8 days, depends on the mouse strain.
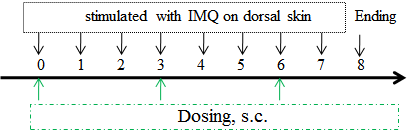
Clinical assessment
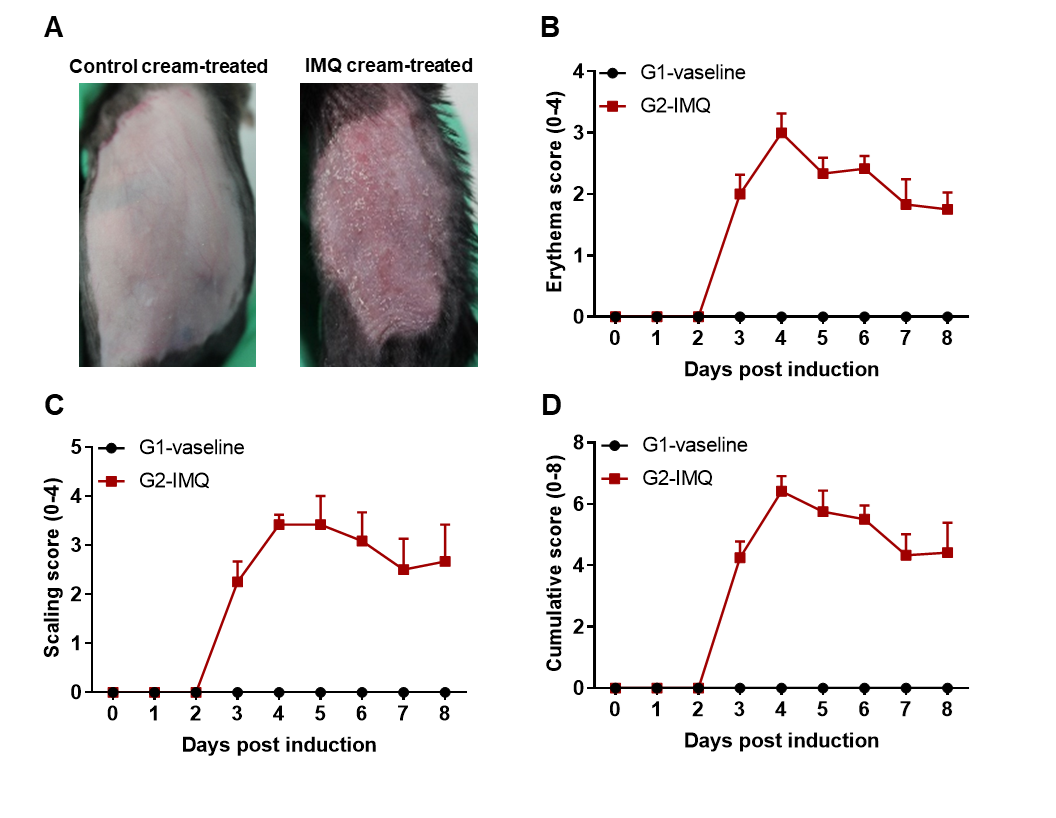
IMQ-induced skin inflammation in mice phenotypically resembles psoriasis. (A) Phenotypical presentation of mouse dorsal skin after 4 days of treatment. (B-D) Erythema and scaling score of the dorsal was scored daily on a scale from 0 to 4. Additionally, the cumulative score (erythema plus scaling) is depicted. Results indicated that structural features of IMQ-induced skin inflammation was visible and increased in severity up to Day 4 then decreased. Mice shaved and treated daily with control cream did not show any sign of inflammation.
Pathological analysis
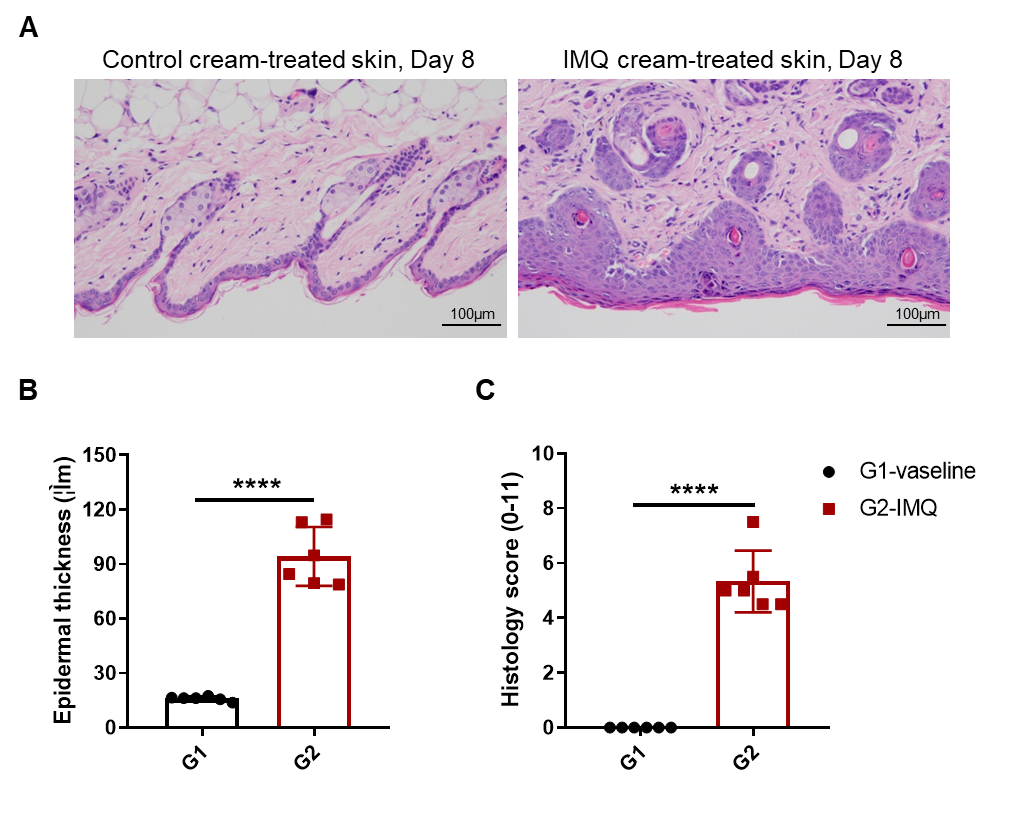
IMQ induces keratinocyte proliferation and inflammatory cell infiltration. (A) H&E staining of the dorsal skin. (B) Epidermal thickness measurement. (C) Histologic score. The epidermal thickness of mice in the IMQ applicated group was significantly increased compared with the control group, and pathological observations indicate that IMQ caused psoriasiform skin lesions and inflammation.
Efficacy Validation on Psoriasis (wild-type C57BL/6) Mouse Models for Dexamethasone
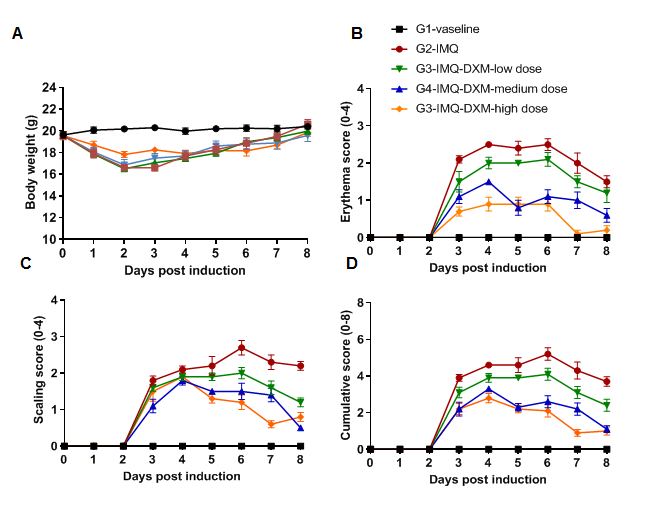
Dose dependent effects of Dexamethasone (DXM) on keratinocyte proliferation and inflammatory cell infiltration in IMQ induced C57BL/6 psoriasis mice. Dorsal skin was collected at the endpoint and stained with Hematoxylin and eosin (H&E). (A-E) H&E staining of the back skin. (F) Epidermal thickness of the mice. (G) Histological changes were scored on a scale from 0 to 11. Results indicated that IMQ cream significantly induced psoriasis-like skin lesions. Therapeutic effects of DXM on psoriasis-like skin lesions in C57BL/6 mice were dose dependent. This model could be used to evaluate the drug efficacy for treating psoriasis.
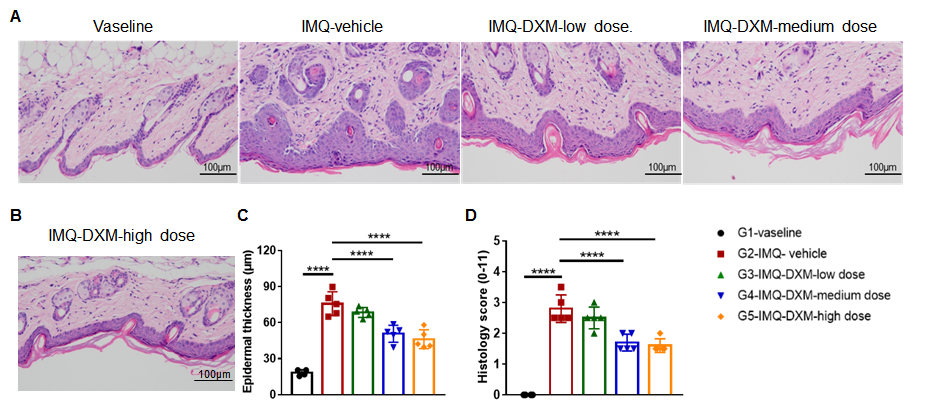
Dexamethasone inhibited keratinocyte proliferation and inflammatory cell infiltration in the C57BL/6 mouse psoriasis model. (A,B) H&E staining of dorsal skin. (C) Mouse epidermal thickness. (D) Histopathology score. The results indicate that dexamethasone prevented psoriasis-like injury and skin inflammation in C57BL/6 mice in a dose-dependent manner.
Efficacy Validation on Psoriasis (B-hIL17A/hIL17F mice) Mouse Models for Bimekizumab (in-house)
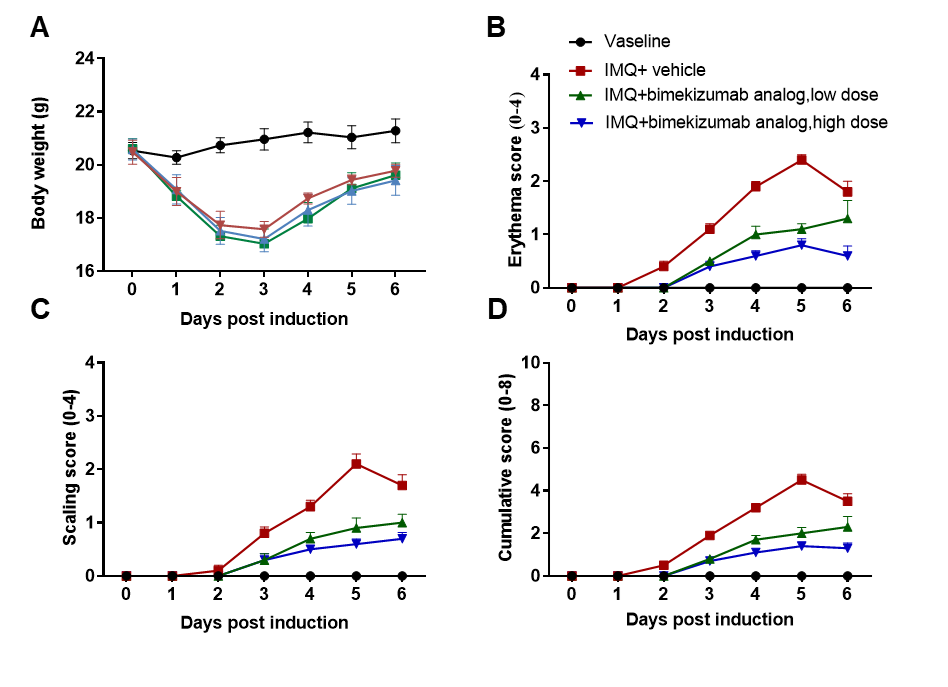
Bimekizumab (synthesized in-house), an anti-human IL-17A and IL-17F antibody drug, prevented psoriasis-like lesions in humanized B-hIL17A/hILl7F mice in a dose-dependent manner. (A) Body weight change during treatment. (B-D) Erythema and scaling score of the dorsal was scored daily on a scale from 0 to 4. Additionally, the cumulative score (erythema plus scaling) is depicted.
Pathological analysis
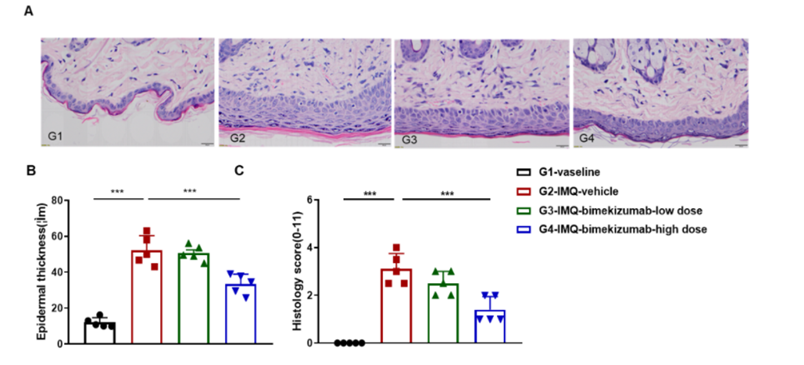
Dose dependent effects of antibody on keratinocyte proliferation and inflammatory cell infiltration in IMQ induced psoriasis-like skin lesions in B-hIL17A/hIL17F mice.
dorsal skin was collected at the endpoint and stained with Hematoxylin and eosin (H&E). (A) H&E staining of the dorsal skin of mice. (B) Epidermal thickness of the mice. (C) Histological changes were scored on a scale from 0 to 11. Results indicated that bimekizumab (in house) significantly reduced psoriasis-like skin lesions in B-hIL17A/hIL17F mice, confirming that B-hIL17A/hIL17F mice is a powerful model for in vivo evaluation of anti-human IL17A and IL17F antibody.
Product list
|
Product name |
Product number |
|
B-hIL17A mice |
110053 |
| B-hIL17A/hIL17F mice | 120554 |
|
B-hTNFA/hIL17A mice |
120548 |
|
B-hIL23A/hIL12B mice |
120553 |
|
B-hCCR6 mice |
110853 |
References
1. van der Fits, L. et al. Imiquimod-induced psoriasis-like skin inflammation in mice is mediated via the IL-23/IL-17 axis. J Immunol 182, 5836-5845.
2. Schuler, R. et al. Antagonization of IL-17A Attenuates Skin Inflammation and Vascular Dysfunction in Mouse Models of







